Quick Summary: This technical guide covers everything biomedical engineers need to know about endoscope biopsy channel replacement — from recognizing damage signs and choosing the right material (PTFE vs alternatives) to sizing by brand/model and reducing costs with quality compatible parts.
In This Guide
- What Is a Biopsy Channel and How Does It Work?
- Signs Your Biopsy Channel Needs Replacement
- PTFE vs Standard Biopsy Channels: Material Comparison
- Biopsy Channel Sizes by Brand and Model
- Step-by-Step Replacement Process Overview
- OEM vs Compatible Replacement Channels: Cost and Quality
- How to Extend Biopsy Channel Lifespan
- Frequently Asked Questions
The biopsy channel is one of the most frequently replaced components in flexible endoscopes. Studies using borescope inspection reveal that 98.5% of endoscopes show superficial scratches in their working channels, and 76% have scratches with adherent peel. With channel damage directly linked to bacterial contamination and infection risk, knowing when and how to replace biopsy channels is critical for patient safety and equipment longevity.
This guide provides biomedical engineers, endoscope repair technicians, and hospital equipment managers with comprehensive technical information on biopsy channel replacement, from damage assessment to material selection and cost analysis.
What Is a Biopsy Channel and How Does It Work?
The biopsy channel, also called the working channel or instrument channel, is a hollow tube running the length of the endoscope insertion tube. It serves multiple critical functions during endoscopic procedures.
This channel allows passage of instruments such as biopsy forceps, snares, cytology brushes, and injection needles. It also functions as a suction channel for removing fluids, blood, and debris during procedures. The channel’s inner diameter determines which instruments can be used and influences suction efficiency.
Most biopsy channels are constructed from polytetrafluoroethylene (PTFE), commonly known as Teflon. PTFE offers a smooth, low-friction surface that facilitates instrument passage and resists chemical degradation from disinfectants. However, the material’s surface degrades with use, becoming rougher and more prone to bacterial colonization over time.
The channel runs from the biopsy port on the control body through the insertion tube to the distal tip opening. This path subjects it to mechanical stress from instrument passes, chemical exposure during reprocessing, and physical stress from scope angulation.
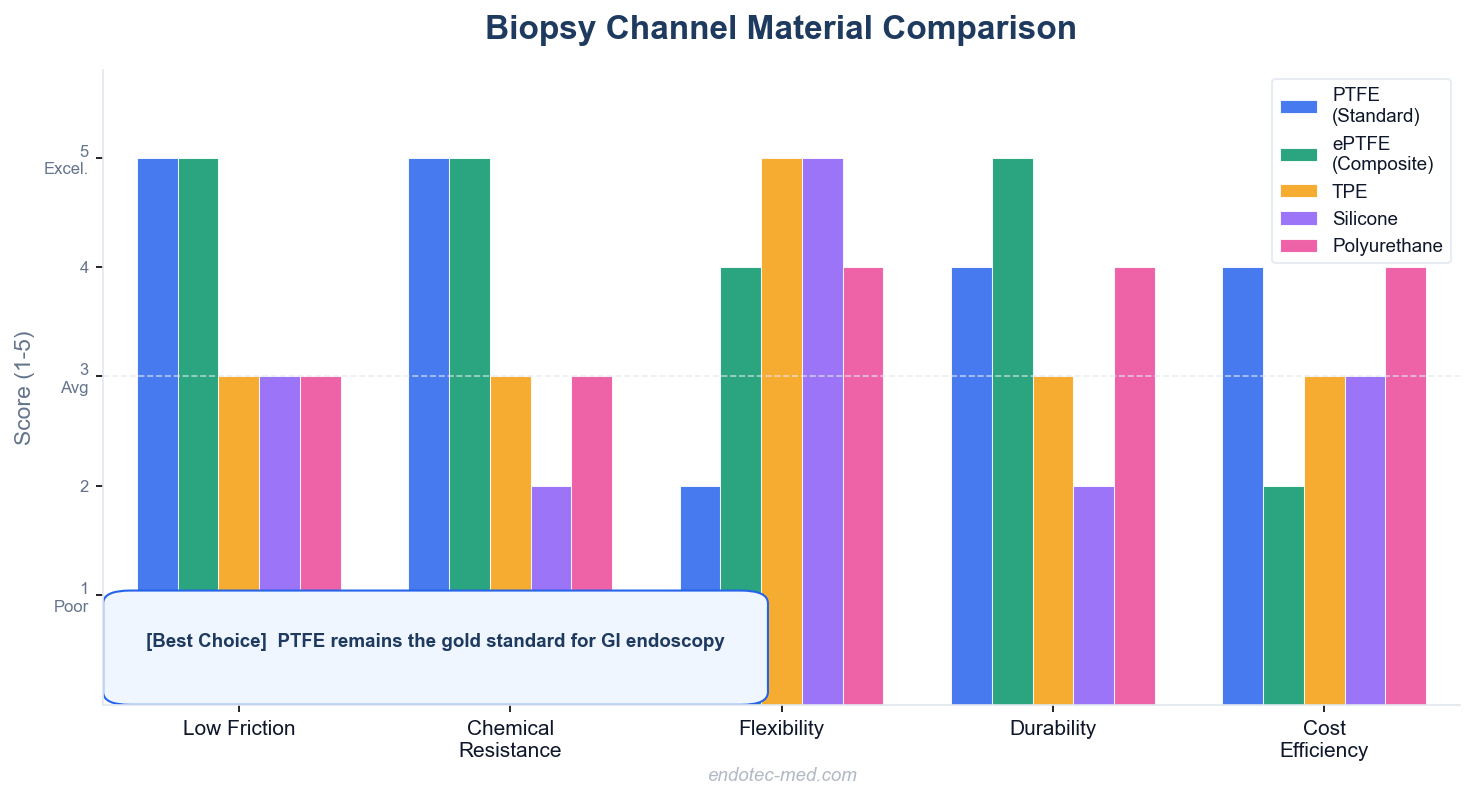
Comparison of biopsy channel materials: PTFE, ePTFE, TPE, Silicone, and Polyurethane
Signs Your Biopsy Channel Needs Replacement
Recognizing channel damage early prevents procedure complications and reduces infection risk. Here are six definitive signs that indicate biopsy channel replacement is necessary.
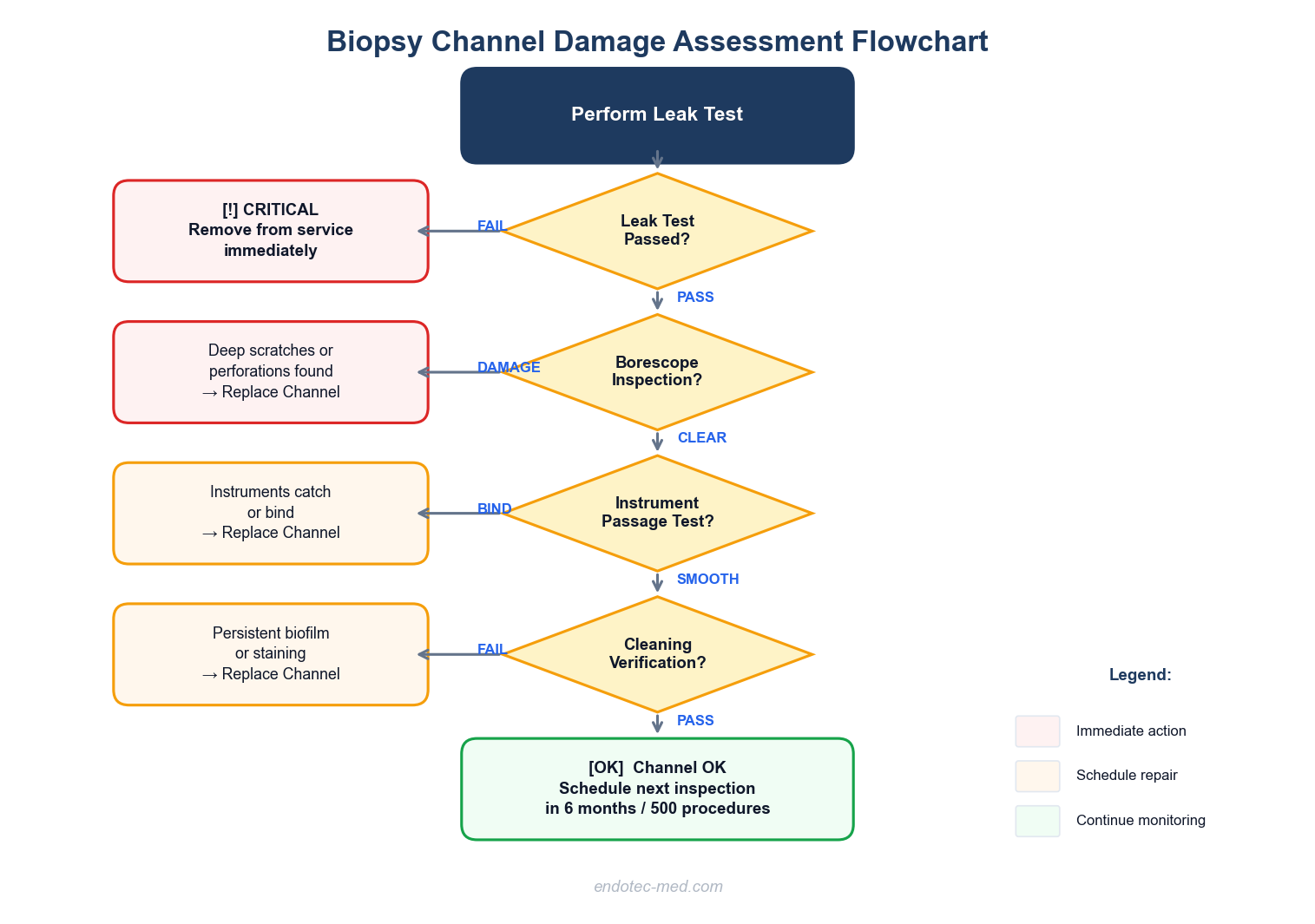
Decision flowchart: When to replace your endoscope biopsy channel
1. Visual damage detected during borescope inspection is the most reliable indicator. Research shows that 85% of stored endoscopes have stains and grooves in their channels, 69% contain moisture, and 46% have debris. A borescope inspection revealing deep scratches, perforations, or significant surface irregularities requires immediate channel replacement.
2. Difficulty passing instruments through the channel suggests internal damage or narrowing. If standard instruments that previously passed smoothly now catch or bind, the channel lining may be torn, kinked, or collapsed. Surface roughness increases 3.2-fold after 500 forceps passes through PTFE channels, creating resistance that technicians can feel.
3. Failed leak tests are a critical red flag. A positive leak test indicates the channel has a breach that allows fluid ingress, which can damage internal electronics and create biofilm reservoirs. Any scope failing a leak test should be immediately removed from service.
Warning: A failed leak test means the scope must be immediately removed from clinical use. Continued use risks fluid invasion into internal electronics, escalating a $975 channel replacement into a multi-thousand dollar complete overhaul.
4. Persistent biofilm or staining despite proper reprocessing suggests surface damage that harbors contamination. Research confirms that used biopsy channels have 1.5 times greater surface roughness than new channels, and this increased roughness correlates with higher bacterial attachment. If cleaning verification consistently fails in the channel, replacement is necessary.
5. Channel kinking or collapse visible externally indicates structural failure. This typically occurs in the bending section where repeated angulation overstresses the channel material. Kinking restricts instrument passage and creates cleaning dead spaces.
6. Discoloration, cloudiness, or opacity of the channel material visible during inspection signals chemical degradation or material breakdown. PTFE channels should remain clear or white; yellowing, browning, or opacity indicates the material has been compromised and should be replaced.
PTFE vs Standard Biopsy Channels: Material Comparison
Understanding material properties helps technicians select the appropriate replacement channel for each application. While PTFE remains the industry standard, alternative materials and composite constructions offer specific advantages.
PTFE (Polytetrafluoroethylene) Channels
PTFE, known commercially as Teflon, dominates the endoscope channel market for valid reasons. Its ultra-smooth, non-stick surface minimizes friction during instrument passage and resists adherent blood, tissue, and mucus. The material’s exceptional chemical resistance withstands repeated exposure to high-level disinfectants and enzymatic cleaners without degrading.
PTFE’s biocompatibility ensures it’s safe for prolonged contact with bodily tissues and fluids. The material won’t leach harmful substances or cause allergic reactions. Its low coefficient of friction reduces wear on instruments passing through the channel.
However, PTFE has limitations. It lacks flexibility compared to elastomeric materials, making it prone to kinking under tight bends. The surface degrades with repeated instrument passes, developing scratches that increase surface roughness by 1.5 to 3.2 times over its service life.
ePTFE (Expanded PTFE) Composite Channels
Advanced manufacturers now offer composite channels combining PTFE with expanded PTFE (ePTFE). The ePTFE layer features a microporous structure of nodes interconnected by fibrils, providing enhanced flexibility. Some designs incorporate circumferential reinforcement to resist kinking while maintaining the low-friction PTFE lumen.
These composite constructions reduce installation friction during manufacturing and improve kink resistance in the bending section. The trade-off is typically higher cost compared to standard PTFE channels.
Alternative Materials: TPE, Silicone, and Polyurethane
Thermoplastic elastomers (TPE) offer superior flexibility and bend without breaking. They’re used in applications requiring exceptional angulation tolerance. However, TPE generally has higher friction than PTFE and may require coating for optimal instrument passage.
Silicone channels provide excellent flexibility and biocompatibility but have limited chemical resistance compared to PTFE. They’re primarily used in specialized applications rather than standard GI endoscopy.
Polyurethane (PU) balances flexibility, durability, and cost-effectiveness. It offers excellent abrasion resistance but requires careful selection of compatible disinfectants.
When to Choose Each Material
For standard GI endoscopes undergoing high-level disinfection with multiple instrument passes per procedure, PTFE remains the optimal choice. Its chemical resistance and low friction outweigh its flexibility limitations in these applications.
For endoscopes with extreme bending requirements or those experiencing repeated kinking failures, ePTFE composite channels justify their additional cost through improved durability. For specialized applications with unique requirements, consult with your biopsy channel supplier to determine the optimal material specification.
Pro Tip: When ordering replacement channels, always specify both the material type AND the exact model number. A “PTFE biopsy channel” without a model specification may not fit your endoscope.
Biopsy Channel Sizes by Brand and Model
Selecting the correct channel diameter is critical for compatibility. Endoscope working channels range from 2.0 mm in ultra-slim gastroscopes to 4.2 mm in therapeutic colonoscopes.
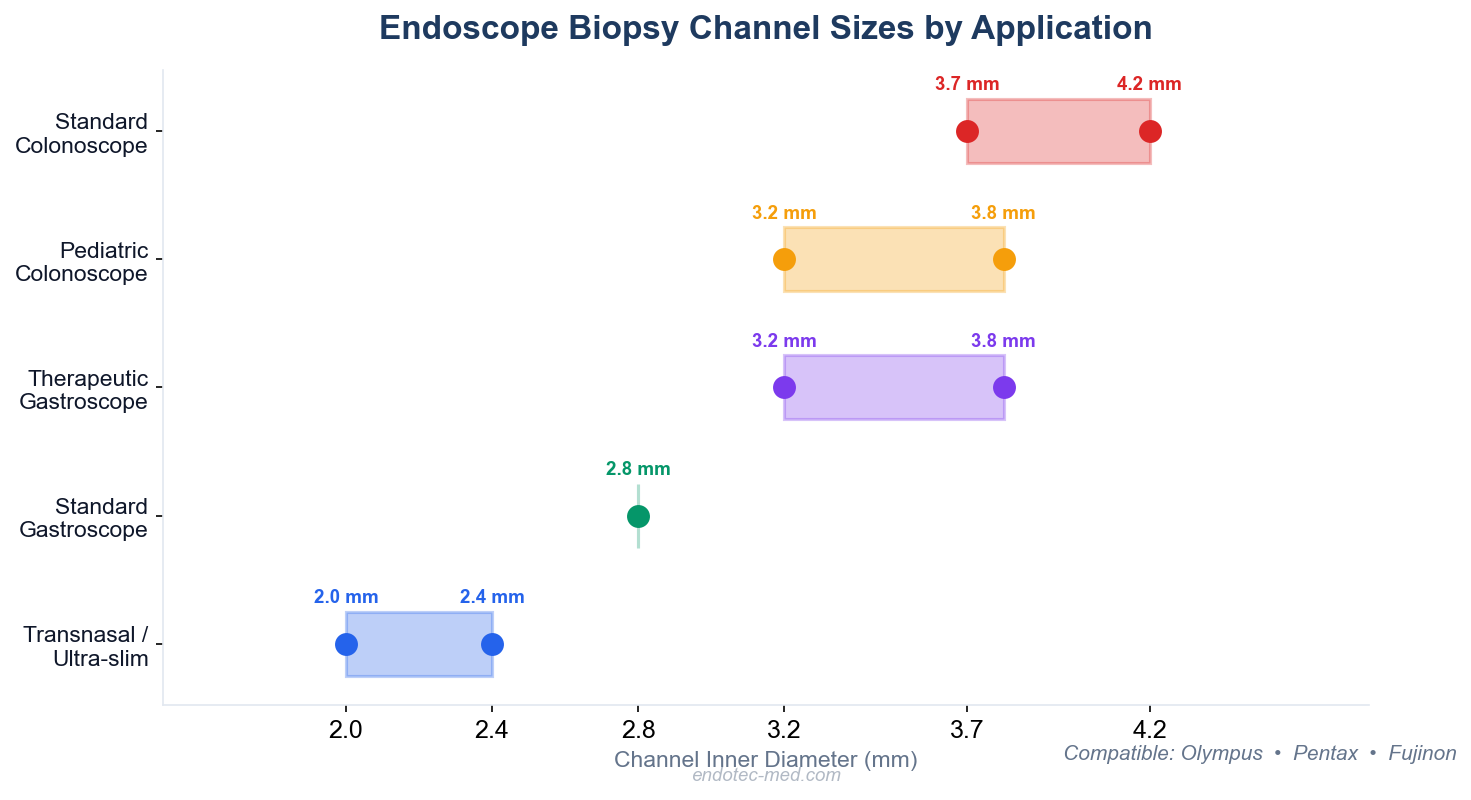
Quick reference: biopsy channel sizes by endoscope application type
Olympus Endoscope Channel Specifications
| Model | Type | Insertion Tube Diameter | Biopsy Channel Diameter |
|---|---|---|---|
| GIF-XP190N | Ultra-slim Gastroscope | 5.4 mm | 2.2 mm |
| GIF-H190 | Standard Gastroscope | 9.9 mm | 2.8 mm |
| GIF-Q260 | Video Gastroscope | 9.1 mm | 2.8 mm |
| PCF-H180AL | Pediatric Colonoscope | 11.3 mm | 3.2 mm |
| CF-H180AL | Standard Colonoscope | 12.8 mm | 3.7 mm |
| CF-HQ190 | HD Colonoscope | 13.2 mm | 3.7 mm |
Olympus typically uses 2.8 mm channels for standard gastroscopes and 3.7 mm for colonoscopes. The ultra-slim GIF-XP190N features a 2.2 mm channel suitable for transnasal procedures.
Pentax Medical Channel Specifications
| Model | Type | Insertion Tube Diameter | Working Channel Diameter |
|---|---|---|---|
| EG-3490K | Therapeutic Gastroscope | 11.6 mm | 3.8 mm |
| EG-3830TK | Dual-Channel Gastroscope | 12.8 mm | 3.8 mm + 2.8 mm |
| EC-3470LK | Pediatric Colonoscope | 11.6 mm | 3.8 mm |
| EC-3890LK | Large-Channel Colonoscope | 13.2 mm | 4.2 mm |
Pentax tends toward larger working channels, with 3.8 mm standard across many models and 4.2 mm in their large-channel colonoscopes.
Fujifilm (Fujinon) Channel Specifications
| Model | Type | Distal Tip Diameter | Working Channel Diameter |
|---|---|---|---|
| EG-740N | Ultra-slim Gastroscope | 5.8 mm | 2.4 mm |
| EG-840T | Therapeutic Gastroscope | 9.8 mm | 3.2 mm |
| EG-840TP | Slim Therapeutic Gastroscope | 7.9 mm | 3.2 mm |
| EI-740D/S | Dual-Channel Endoscope | — | 3.7 mm + 3.2 mm |
Standard Channel Sizes by Application
| Application | Typical Channel Diameter |
|---|---|
| Transnasal / Ultra-slim gastroscopes | 2.0-2.4 mm |
| Standard diagnostic gastroscopes | 2.8 mm |
| Therapeutic gastroscopes | 3.2-3.8 mm |
| Pediatric colonoscopes | 3.2-3.8 mm |
| Standard colonoscopes | 3.7-4.2 mm |
| Dual-channel (secondary) | 2.8-3.2 mm |
When ordering replacement channels, verify both the inner and outer diameter specifications. The outer diameter must fit within the insertion tube assembly, while the inner diameter determines instrument compatibility. Consult your endoscope’s service manual or contact us for model-specific specifications.
Step-by-Step Replacement Process Overview
Biopsy channel replacement requires specialized tools and training on the specific endoscope model. This overview outlines the general process; always consult manufacturer service manuals for model-specific procedures.
Pre-Replacement Assessment
Perform a comprehensive leak test to confirm channel damage and identify the leak location. Document the failure with borescope inspection photos. Verify the correct replacement channel part number from the service manual or by measuring the damaged channel’s inner and outer diameter.
Disassembly Sequence
Remove the control body from the insertion tube. This typically involves disconnecting electrical connections, air/water connections, and removing securing screws. Label all connections for proper reassembly. Remove the distal tip assembly, noting the orientation of components and o-rings.
Thread the damaged channel out from the distal end, carefully noting its path through the insertion tube and bending section. Take photos of the channel routing to ensure correct replacement installation.
Channel Installation
Thread the new channel through from the distal end, following the exact path of the original. Ensure the channel isn’t twisted and passes smoothly through the bending section without kinking. Secure the distal end with appropriate adhesive, ensuring a fluid-tight seal. Allow adhesive to cure per manufacturer specifications.
Route the proximal end to the biopsy port and secure with adhesive. Verify the channel protrudes the correct distance at both ends. Reassemble the distal tip, carefully positioning all o-rings and seals.
Post-Replacement Verification
Perform a complete leak test on the newly installed channel. Test at the manufacturer’s specified pressure (typically 25 kPa or 3.6 psi) for the recommended duration. Inspect for bubbles indicating leaks at adhesive joints.
Perform instrument passage tests with appropriately sized biopsy forceps and brushes. Verify smooth passage without catching or resistance. Test suction function to confirm proper channel patency and seal integrity.
Clean and high-level disinfect the entire endoscope per AAMI ST91 guidelines before returning to service. Document the replacement in the scope’s service history with date, channel specifications, and post-repair test results.

Quality inspection of endoscope insertion tube assembly during service
OEM vs Compatible Replacement Channels: Cost and Quality Analysis
Biomedical departments face a critical decision when sourcing replacement channels: original equipment manufacturer (OEM) parts versus compatible alternatives. Understanding the trade-offs informs cost-effective procurement strategies.
OEM Channel Advantages
OEM channels offer guaranteed compatibility with manufacturer specifications. They’re manufactured to the exact tolerances and material specifications designed for your specific endoscope model. OEM parts typically come with manufacturer support, including detailed installation instructions and warranty coverage.
However, OEM channels command premium pricing. A complete overhaul from manufacturers can run into thousands or tens of thousands of dollars, with channel replacement being a significant component of that cost.
Compatible Replacement Channels
Independent manufacturers like Endotec produce compatible replacement channels at substantially lower costs. Research indicates that third-party repair services charge approximately $975 for biopsy channel replacement, representing 50-60% savings compared to OEM service prices.
Quality compatible channels use the same PTFE material and manufacturing processes as OEM parts. Reputable suppliers provide dimensional specifications, material certifications, and compatibility charts for major brands including Olympus, Pentax, and Fujifilm.
The risk with compatible parts lies in sourcing from unverified suppliers. Low-quality channels may use inferior materials, have dimensional inaccuracies, or lack proper quality control.
Cost-Benefit Analysis
Consider a facility performing 10,000 endoscopic procedures annually with an endoscope service budget of $152,000. One documented case study showed a facility reduced annual repair spending from $52,000 to $30,000-40,000 by bringing some repairs in-house using compatible parts, even as procedure volume quadrupled.
Key Takeaway: For facilities with experienced biomedical staff and established quality control procedures, high-quality compatible channels from reputable suppliers like Endotec represent excellent value — achieving 50-60% cost savings without compromising performance.
How to Extend Biopsy Channel Lifespan
Preventive maintenance and proper handling significantly extend channel service life, reducing replacement frequency and associated costs.
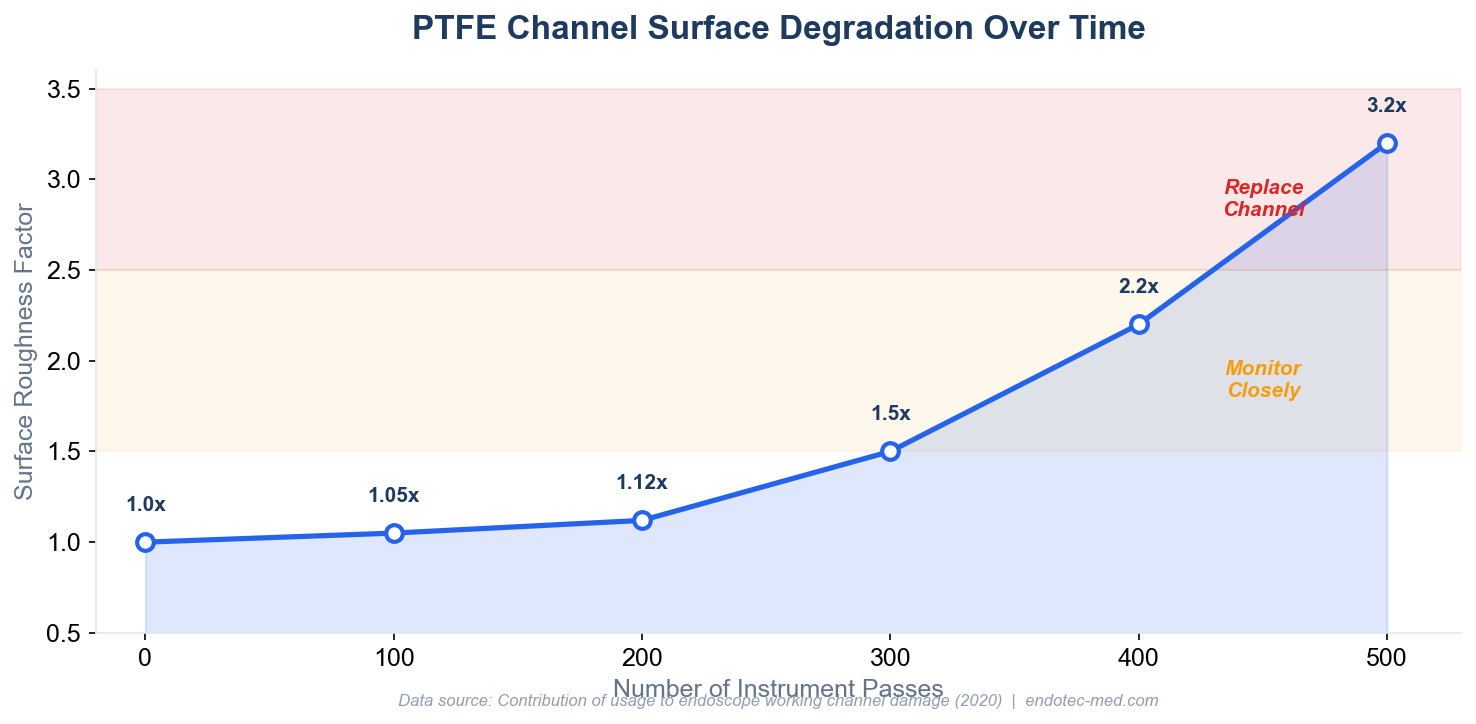
How PTFE surface roughness degrades with repeated forceps passes
Follow AAMI ST91 Reprocessing Guidelines
The 2021 AAMI ST91 standard provides comprehensive guidance for endoscope reprocessing that directly impacts channel longevity. Aspirate enzymatic detergent through all channels immediately at point of use. This prevents protein fixation that makes cleaning difficult and creates channel surface irregularities.
During manual cleaning, brush channels with appropriately sized cleaning brushes that aren’t worn or damaged. Worn brushes with protruding bristles or metal can scratch the PTFE surface, accelerating degradation.
Ensure proper drying per ST91 recommendations: minimum 10 minutes using pressure-regulated forced instrument air or HEPA-filtered air. Moisture retention in channels creates an environment for biofilm formation.
Use Compatible, Well-Maintained Instruments
Verify all instruments are compatible with your endoscope’s channel diameter. Using oversized instruments stresses the channel and causes mechanical damage. Research demonstrates that surface roughness increases progressively with instrument passes: 1.05-fold after 100 passes, 1.12-fold after 200 passes, and 3.2-fold after 500 passes.
Implement Rigorous Leak Testing Protocols
Perform leak tests before every reprocessing cycle. Detecting channel leaks early prevents fluid invasion into the insertion tube, which would escalate repair costs from a $975 channel replacement to a multi-thousand dollar complete overhaul.
Establish a Preventive Maintenance Schedule
Implement regular borescope inspections to detect early channel damage before it progresses to leaks. Schedule inspections every six months or after every 500 procedures, whichever comes first for high-use scopes. Document findings with photos to track degradation trends.
For more information on maintenance best practices, refer to our comprehensive guide to endoscope biopsy channels.
Frequently Asked Questions
How often should endoscope biopsy channels be replaced?
Replacement frequency depends on usage intensity and maintenance practices rather than a fixed schedule. Replace channels when leak tests fail, borescope inspection reveals significant damage, or when instruments no longer pass smoothly. High-volume facilities may need replacement every 2-4 years for heavily used scopes, while properly maintained low-volume endoscopes may last significantly longer.
Can I replace a biopsy channel in-house or must I send the scope to the manufacturer?
In-house replacement is feasible for facilities with trained biomedical technicians and appropriate tools. Many facilities perform channel replacements in-house using compatible parts from suppliers like Endotec, achieving 50-60% cost savings compared to OEM service. However, complex endoscope models or scopes under manufacturer warranty may require OEM service.
What’s the difference between a biopsy channel and a suction channel?
In most flexible endoscopes, the biopsy channel and suction channel are the same structure, serving dual purposes. Some specialized endoscopes feature separate suction channels. Dual-channel therapeutic endoscopes like the Fujifilm EI-740D/S have two separate working channels (3.7 mm + 3.2 mm), each capable of instrument passage and suction independently.
Will using compatible replacement channels void my endoscope warranty?
Warranty implications vary by manufacturer and contract terms. Many manufacturers stipulate that using non-OEM parts or unauthorized service providers voids warranty coverage. However, endoscope warranties are typically limited (1-2 years), and many repairs occur on equipment outside the warranty period. For out-of-warranty scopes, using quality compatible channels presents minimal risk and substantial cost savings.
What happens if I delay replacing a damaged biopsy channel?
Delaying channel replacement creates escalating risks and costs. A damaged channel harbors bacterial biofilms in scratches and surface irregularities. Continued use of a leaking channel allows fluid invasion into the insertion tube, corroding internal components and potentially damaging electronics. This escalates repair costs from approximately $975 for isolated channel replacement to thousands of dollars for complete scope overhaul.
Conclusion
Endoscope biopsy channel replacement is a critical maintenance procedure that directly impacts patient safety, procedural success rates, and long-term equipment costs. Implementing systematic inspection protocols catches damage early, when channel replacement remains straightforward and cost-effective.
Sourcing quality compatible channels from reputable suppliers like Endotec achieves 50-60% cost savings without compromising performance. Following AAMI ST91 reprocessing guidelines, using well-maintained instruments, and training staff on proper handling extends channel service life significantly.
Ready to source quality replacement biopsy channels for your endoscope fleet? Contact our technical team for model-specific specifications, compatibility verification, and volume pricing.
Sources
- Contribution of usage to endoscope working channel damage and bacterial contamination
- Scoping the scope: endoscopic evaluation of endoscope working channels
- ANSI/AAMI ST91:2021 Flexible and semi-rigid endoscope processing
- Multisociety guideline on reprocessing flexible GI endoscopes
- Global Endoscopy Solutions Repair Prices
- Rein in Endoscope Service Costs

